top of page
iPharmaCenter

For sponsored articles, link insertions, and advertisements, please write to info@ipharmacenter.com
For most cost-effective consulting services:
Germany G-BA Assessment Outcomes
Rephrase with Ginger (Cmd+⌥+E)

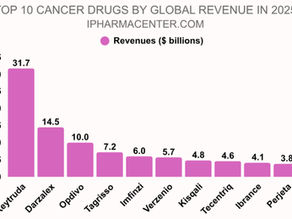
Best-Selling Cancer Drugs 2025: Top 10 by Revenue
BEST SELLING CANCER DRUGS | TOP ONCOLOGY DRUGS | 2025 BEST SELLING CANCER DRUGS 2025 Rank Brand Company Indications Revenues $ billion 1 Keytruda Merck Melanoma, NSCLC, HNSCC, Hodgkin Lymphoma, Urothelial Cancer, RCC, HCC and several other cancers $31.7 2 Darzalex Johnson and Johnson Multiple myeloma $14.5 3 Opdivo Bristol Myers Squibb Melanoma, NSCLC, Hodgkin Lymphoma, Urothelial Cancer, RCC, HCC and several other cancers $10.0 4 Tagrisso AstraZeneca EGFR positive NSCLC $7.2


EULAR Annual Meeting in London, UK | 2026 | News | Updates | iPharmaCenter
JOHNSON AND JOHNSON Nipocalimab Shows Stronger Phase 2 Signal in Autoantibody-High Sjögren’s Disease Nipocalimab is attracting attention at EULAR 2026 as a potential new option for patients with Sjögren’s disease who have a high autoantibody burden. Fresh Phase 2 data from the DAHLIAS study suggest that the patients with the most pronounced serologic activity may be the ones who gain the greatest clinical benefit. Signal in patients with high autoantibody and IgG levels DAHLI
American Society of Clinical Oncology (ASCO) Annual Meeting | Chicago | 2026
ASTRAZENECA STRIDE Plus Lenvatinib and TACE Cuts Progression Risk by 30% in Embolisation‑Eligible Unresectable HCC Early EMERALD‑3 results indicate that combining dual checkpoint blockade and lenvatinib with TACE can prolong disease control in patients with embolisation‑eligible, unresectable hepatocellular carcinoma, without introducing new safety concerns. How EMERALD‑3 was designed? EMERALD‑3 is a Phase III study enrolling people with unresectable HCC who are suitable fo
bottom of page